
About Propane - Characteristics, Properties and Combustion
Propane is a liquefied petroleum gas that comes out of both oil and gas wells. Propane does not occur naturally though. Raw crude oil or raw natural gas is refined to make different types of petroleum products, one of which is propane. Following it's refinement, propane is stored as a liquid under pressure until utilized, at which point it becomes a gas.
Propane Properties
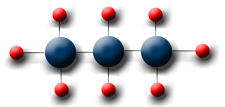
C3H8 - The formula for propane is 3 Carbon molecules and 8 Hydrogen molecules.
| Propane Boiling Point | -44°F |
| Weight of 1 Gallon Liquid Propane* | 4.24 Lbs |
| Specific Gravity of Propane Gas* | 1.52 |
| Specific Gravity of Liquid Propane* | .51 |
| BTU per Gallon of Propane Gas* | 91,547 |
| BTU per Lb. of Propane Gas | 21,591 |
| BTU per Cu Ft of Propane Gas* | 2,516 |
| *at 60 degrees F |
Propane Properties Explained
Propane Boiling Point - Water boils at 212°F meaning that it becomes a gas at this temperature whereas water is still a liquid at 200°F. Propane is a liquid at -50°F and boils at -44°F. In other words, at 10 degrees below zero, propane is well past its boiling point. What does this mean to ordinary people like us? It means that propane is cold enough freeze your skin (and tissue underneath) to the point of severe damage.
Specific Gravity of Propane Gas - One cubic foot of propane weighs .1162 lbs. and one cubic foot of air weighs .07655. Dividing .1162 by .07655 equals 1.52. What does this mean? Propane is heavier than air and will seek the lowest space available.
Specific Gravity of Liquid Propane - One gallon of propane weighs 4.24 lbs. One gallon of water weighs 8.33 lbs. Dividing 4.24 by 8.33 equals .51. This means propane is less dense than water (lighter than water) and will not sink in water, so to speak.
BTU - As defined, a British Thermal Unit is the quantity of heat required to raise the temperature of one pound of water by one degree Fahrenheit. It takes about 143 BTU, or .0016 gallons of propane, to melt one pound of ice (at 32°F). Most all installation requirements and decisions revolve around BTU ratings and demand.
Propane Combustion Characteristics
| Lower Limit of Flammability | 2.15 |
| Upper Limit of Flammability | 9.60 |
| Flash Point | -156°F |
| Ignition Temperature in Air | 920-1020°F |
| Maximum Flame Temperature | 3595°F |
| Octane Number | Over 100 |
Propane, Flammability and Combustion Requirements
In order for propane to burn, ignite or go through combustion, the criteria listed above must be met. Below are explanations of propane gas combustion characteristics.
Propane Limits of Flammability - The lower and upper limits of flammability are the percentages of propane that must be present in an propane/air mixture. This means that between 2.15 and 9.6% of the total propane/air mixture must be propane in order for it to be combustible. If the mixture is 2% propane and 98% air, there will not be combustion. If the mixture 10% propane and 90% air, combustion will not occur. Any percentage of propane in a propane/air mixture between 2.15% and 9.6% will be sufficient for propane to burn. However, an improper air/gas mixture can produce Carbon Monoxide (CO) that is a deadly product of incomplete combustion.
Flash Point - The flash point is the minimum temperature at which propane will burn on its own after having been ignited. This number states that below -156°F, propane will stop burning on it's own. In other words, if the outside air temperature is -155°F, propane will burn on it's own. If the outside air temperature falls to -157°F, propane will no longer burn on it's own. However, if a source of continuous ignition is present, propane will burn below - 156°F.
Ignition Temperate in Air - This number states that propane will ignite if it reaches a temperature between 920-1020°F. If propane is heated up to a temperature between 920 and 1020°F, it will ignite without needing a spark or flame.
Maximum Flame Temperature - A propane flame will not burn hotter than 3595°F.
Propane Octane Number - Without presenting a chemistry lesson, the Octane number of propane being over 100 means that it is a very engine friendly fuel.
See Propane Liquid and Propane Vapor for more detailed information about the physical states of propane.
